In our lab, we employ a number of synthesis techniques to bring our materials to life. We then use a range of characterisation techniques – both here in Birmingham, and at central facilities – in order to understand their fascinating behaviour.
Synthesis

Solid-state synthesis is a widely used method in materials chemistry for the synthesis of powder metal oxides. Measured amounts of reactants are ground together and pressed into a pellet before heating. The grinding and pressing stages are to ensure a small, homogeneous, particle size and to increase the contact area between reactants. Usually the sample has to be fired at very high temperatures (< 2000 oC) to make the final product.
Solvothermal synthesis is another commonly used method, with the benefit over solid-state techniques of requiring much lower reaction temperatures. Here, reactants are gently heated in a solvent, if the solvent is water then this process is known as hydrothermal synthesis. In our lab, we use hydrothermal methods to make materials like barlowite as it mimics the natural crystal growth conditions in Australian quarries.
The synthesis of materials is very complex – there are so many variables to consider for each reaction; reactants, temperature, firing time, solvent, atmosphere… Preparing samples with a known method can be a delicate process, and making new materials can take months of experiment before we get it just right!
Diffraction
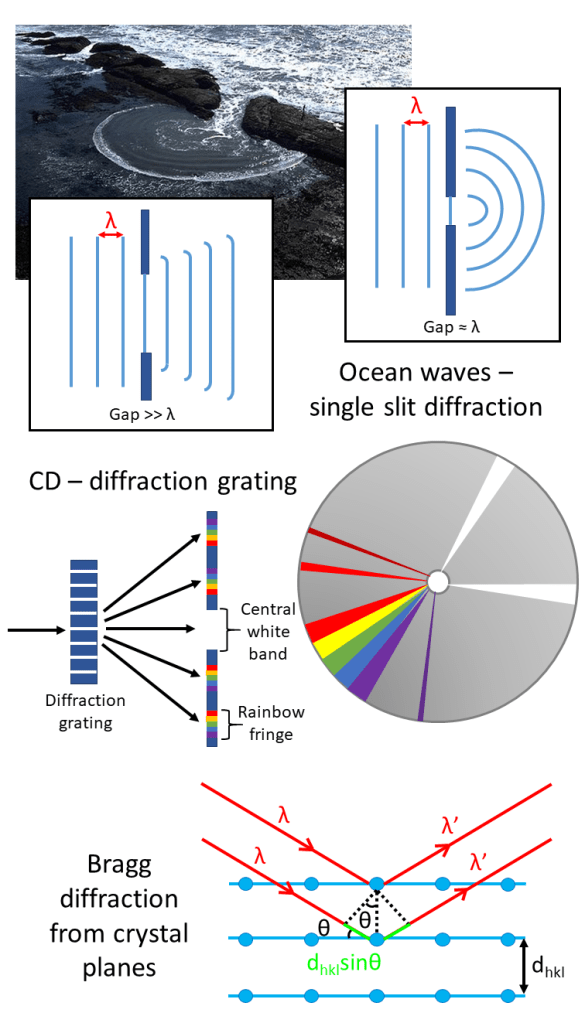
Have you ever wondered why ocean waves form circular patterns when flowing into a harbour? Or why when you tilt a CD it appears to reflect the rainbow? The answer is diffraction – a phenomenon that occurs as waves encounter obstacles in their path.
In the case of ocean waves, the distance between the crest of one wave to the next is known as the wavelength, λ. Diffraction is maximised when λ equals the size of the gap (or slit size) leading to the harbour – the closer the match, the more circular the pattern appears.
For a CD, the closely spaced etchings that give the CD its storage can also act as an optical diffraction grating – splitting white light into its constituent parts.
Diffraction can occur with any kind of wave. In our lab, we use X-rays to determine the structure of the materials we create. The same principle applies, the X-rays paths are disrupted by the crystal planes and the interference effects – whether the waves superimpose or cancel each other out – produce characteristic patterns that we can analyse. This is known as Bragg diffraction and is one of our cornerstone techniques.
Why Use Neutrons?
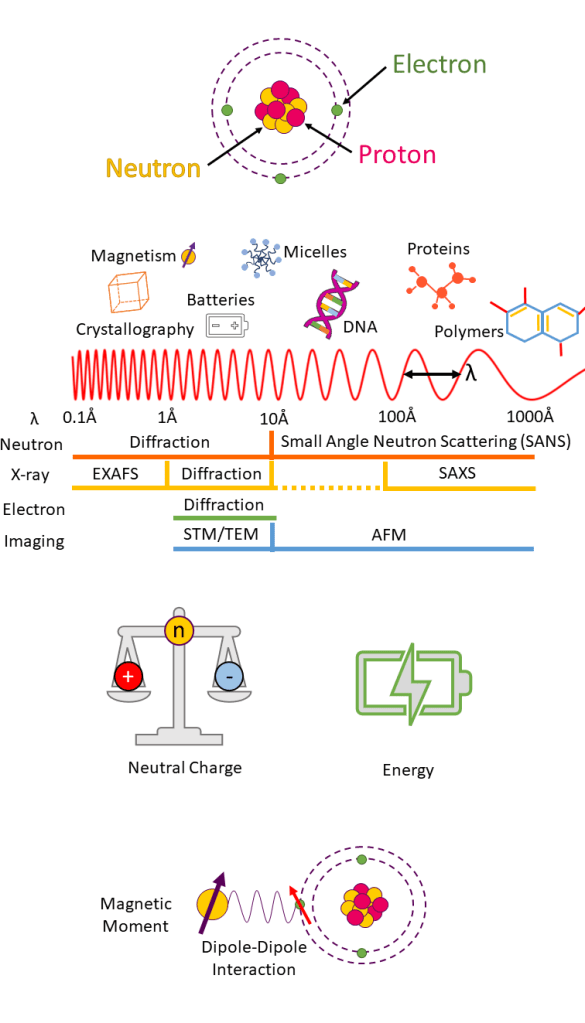
Atoms contain three sub-atomic particles; protons, neutrons and electrons. Protons and neutrons make up an atoms nucleus, and electrons occupy shells around the nucleus.
Some particles, like neutrons and electrons, can experience wave-like properties, and therefore, can also be used in diffraction studies.
Neutron wavelengths range from 0.1 – 1000 Å (1 Å = 10-10 m) which is similar in size to interplanar crystal spacing. Combined with their neutral charge, this allows neutrons to penetrate deep into materials making them an ideal probe of atomic and molecular structures in diffraction studies.
Neutron energies are the same magnitude as excitation energies in crystals allowing a materials dynamics to be probed in inelastic (initial energy ≠ final energy) measurements.
Neutrons also have a magnetic moment and can therefore interact with unpaired electrons in a material. Low-temperature diffraction and polarised scattering techniques allow us to measure a materials magnetic structure.
Muon Spectroscopy
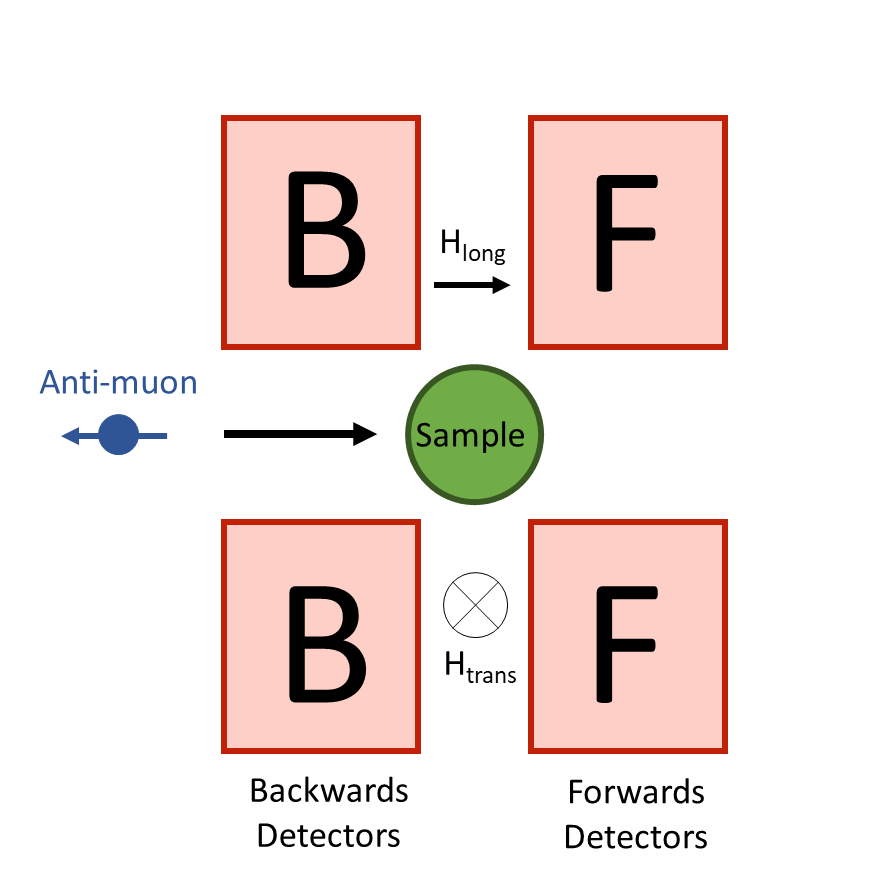
A muon can be thought of as a heavy electron, around 200x the mass, in fact! Like electrons, muons also have a magnetic moment and so can be used to study magnetic materials. We commonly use muon spin rotation and relaxation (μSR) to study the dynamics within our materials.
In μSR, polarised muons are implanted into a sample and come to rest at well-defined sites. Typically, the sample is placed between two sets of detectors, a forward and backwards set. Depending on the type of measurement, a magnetic field can be applied in a transverse or longitudinal direction to the sample. This causes the muon to precess between the banks, which may result in an asymmetric signal if the magnetic moments are fluctuating.
Magnetisation Measurements

Magnetic materials have signatures that tell us what sort of order they have. Of course, in our group we are looking for the absence of these features, or variations that suggest a more complex, possibly disordered ground state.
We measure a materials bulk magnetisation characteristics using a Superconducting QUantum Interference Device (SQUID). Most metallic conductors have electrical resistance, meaning that some of the energy is lost, as heat for example. But in a superconductor, below a critical temperature there is no resistance at all, making these materials very sensitive to small magnetic effects. In a SQUID, a superconducting ring surrounds the sample and an external magnetic field is applied. As the sample moves through the ring, the moments will align according to their inherent magnetic state, which we measure as magnetic susceptibility.
We either record with a direct current (DC) or alternating current (AC).
Where we do this work

Some of these measurements, we can do in-house. But for more in-depth experiments we travel to specialised central facilities, both in the UK and abroad.
ISIS Neutron and Muon Source – the UK’s national neutron facility based at the Harwell campus in Didcot. This is a spallation source where protons travel at very high speeds in a circular ring and when they collide with a tungsten target produce neutrons and muons, that are then used in experiments. Some of the ISIS instruments we have used recently are HRPD, WISH, MuSR and MAPS. We also make use of the Materials Characterisation Laboratory for magnetisation measurements.
Diamond Light Source – the UK’s national synchrotron facility, also based at the Harwell campus. This is a third-generation X-Ray source where X-Rays are 10 billion times brighter than the sun! We recently took our first group trip there to use the I11 beamline for high-resolution structural studies.
Institut Laue-Langevin – a reactor based facility at the EPN campus in Grenoble, France. Nuclear fission reactors are not only used for energy purposes, they can also be used for fundamental science. This can have advantages over pulsed sources like ISIS because they produce a higher, continuous flux of neutrons (amount of neutrons per unit area). We have made use of the ILL’s D7, D20 and D2B diffractometers.
Paul-Scherrer Institute – PSI operates a collection of neutron and X-ray facilities in Switzerland. We have conducted muon experiments on the GPS instrument at the SμS: Swiss Muon Source.
See how we apply these techniques to our science in our research highlights page.

